OUR SOLUTIONS
Don’t move, We’ll Handle Everything
RESS solution
Our Renewable Energy Storage System (RESS) provides highly efficient and safe energy storage solutions for power grids, power systems, and renewable energy facilities.
I&C solution
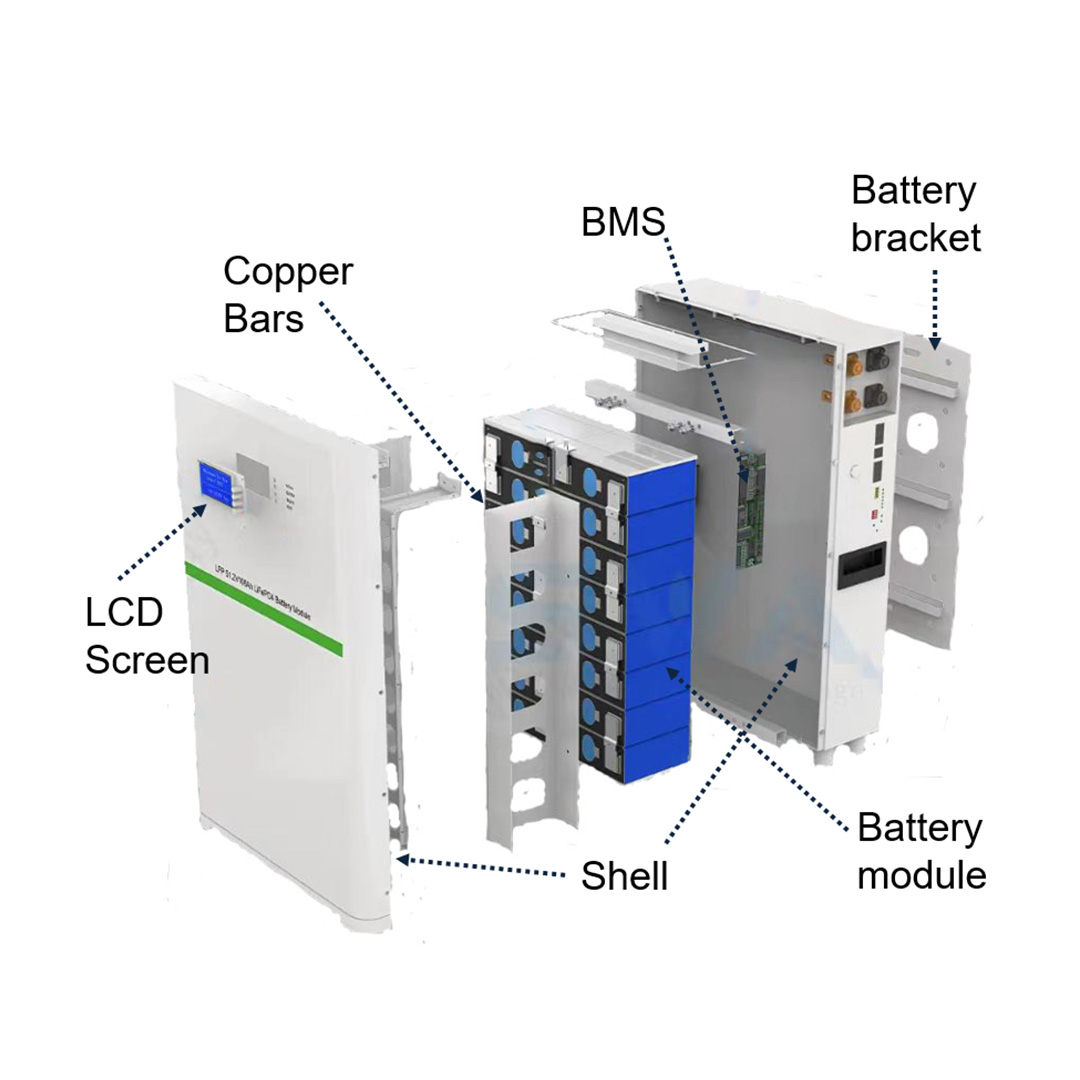
Designed for rugged industrial environments, our energy storage systems feature robust enclosures, advanced thermal management, and intelligent BMS.
Telecom ESS
Our Telecom Energy Storage solution provides reliable and cost-effective backup power for telecommunications infrastructure and
equipment.
UPS Solution
Facing the risks of power outages and voltage fluctuations, our UPS (Uninterruptible Power Supply) provides reliable backup power for critical loads.
DISCOVER WHAT SETS FARAN APART
Why Choose Us?
- Customized Solutions and Support
- Innovative Technology and Products
- Commitment to Sustainability
- Global Leadership and Reliability
QUALIFICATIONS
Successfully passed ls09001,1ATF16949
ISO14001,QHSAS18001, SA8000, all
R&D CAPABILITIE
Rich manufacturing experience
automative cell production line and pack
MANUFACTURING
Obtained more than 20 invention
patents and more than 60 utility model
CHANNELS & NETWORK
Product sold in more than 60 countries
and regons in Asia Pacific, Africa.
Previous slide
Next slide
Excellent battery manufacturer
Our Products
Our happy customers
We are committed to providing you with the best battery solutions

Wilson
Manager UrbanAcres, LLC
★★★★★ 5/5
“I had a great experience working with this company. Their team was knowledgeable, professional, and responsive to all of my needs.”

Angela
Project Manager
★★★★★ 5/5
“This company exceeded my expectations in every way. Their attention to detail and commitment to quality were truly exceptional.”

John Doe
Director
★★★★★ 5/5
“Working with this company was a breeze. They were efficient, communicative, and delivered exactly
what I was looking for.”
0
+
Years Experience
0
/5
Customer satisfaction Rate
0
Professional team members